Market Intel: Glucose Monitor Market Set To Explode As Patients Access Better Devices :: Medtech Insight

FDA Issues Final Orders to Reclassify Blood Lancet Devices into Class II and Class III Devices; Orders Include a PMA Requirement for Class III Blood Lancets - Registrar Corp

CGM accuracy: Contrasting CE marking with the governmental controls of the USA (FDA) and Australia (TGA): A narrative review - Pemberton - 2023 - Diabetes, Obesity and Metabolism - Wiley Online Library

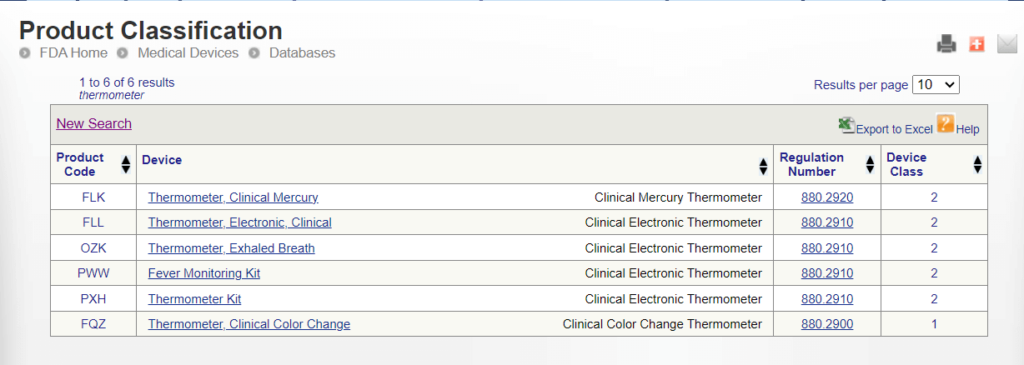
Glucose meters: current regulatory guidance for manufacturers and providers | Medical Laboratory Observer

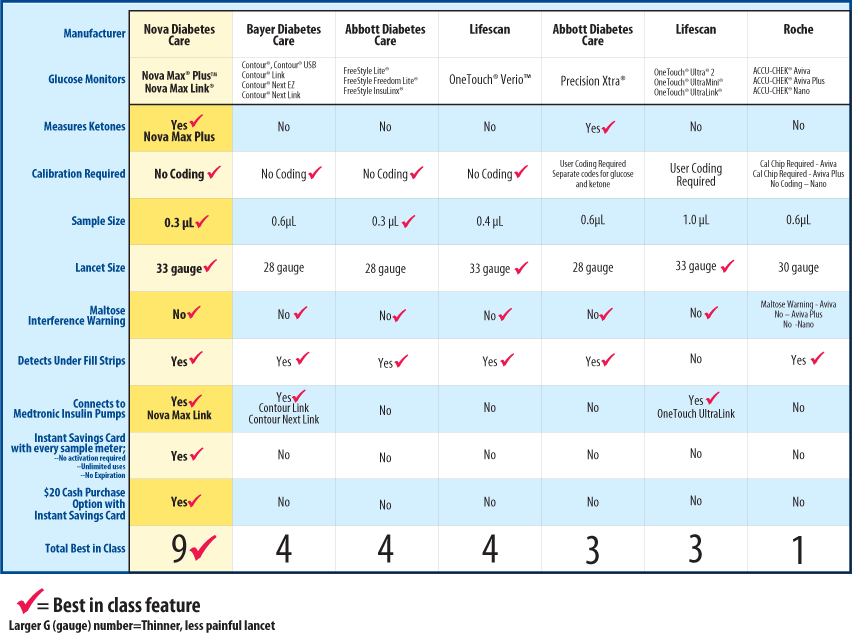
CE FDA ISO Approved Health Care Bluetooth Diabetes Blood Sugar Test Kit Glucose Meter - China Glucometer, Glucose Meter | Made-in-China.com
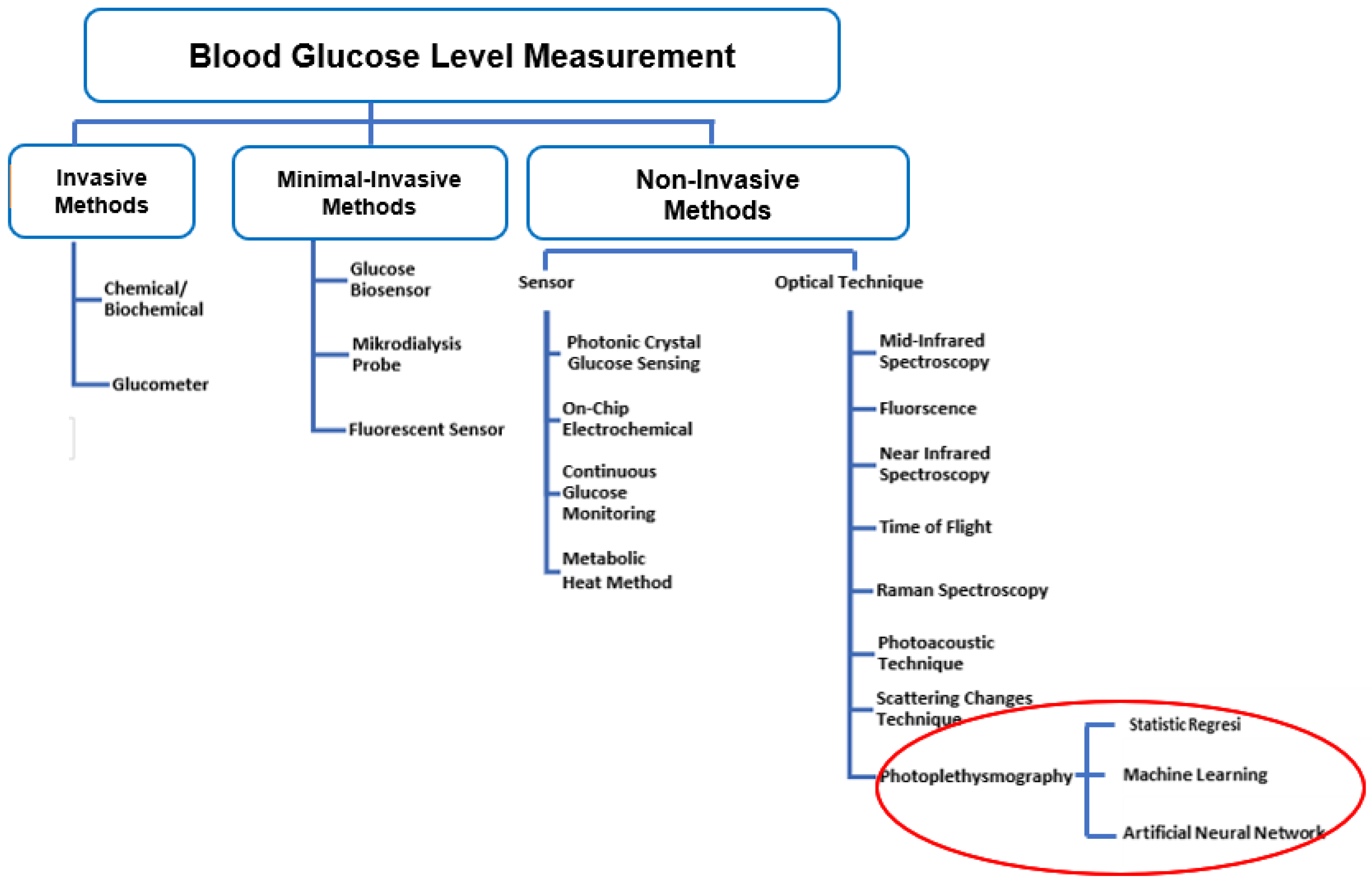
Information | Free Full-Text | Non-Invasive Classification of Blood Glucose Level for Early Detection Diabetes Based on Photoplethysmography Signal

Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf
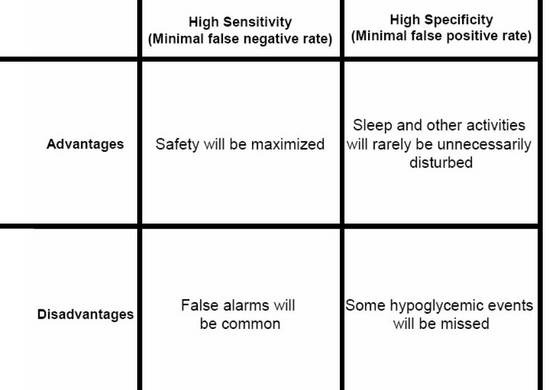
Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf

Glucose meters: current regulatory guidance for manufacturers and providers | Medical Laboratory Observer

Glucose meters: current regulatory guidance for manufacturers and providers | Medical Laboratory Observer
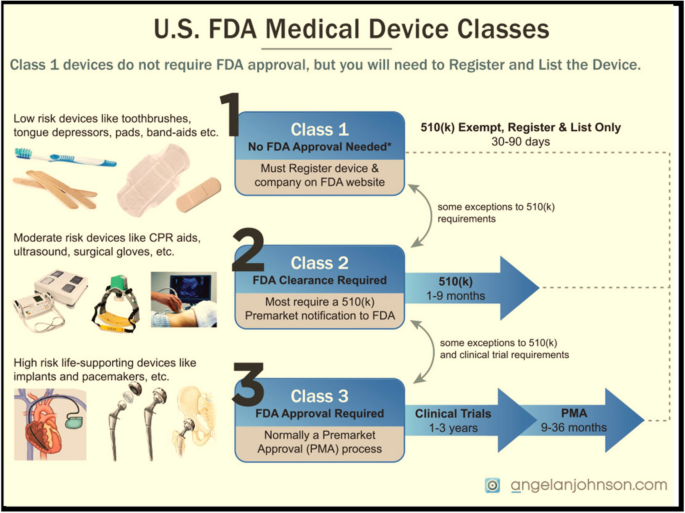
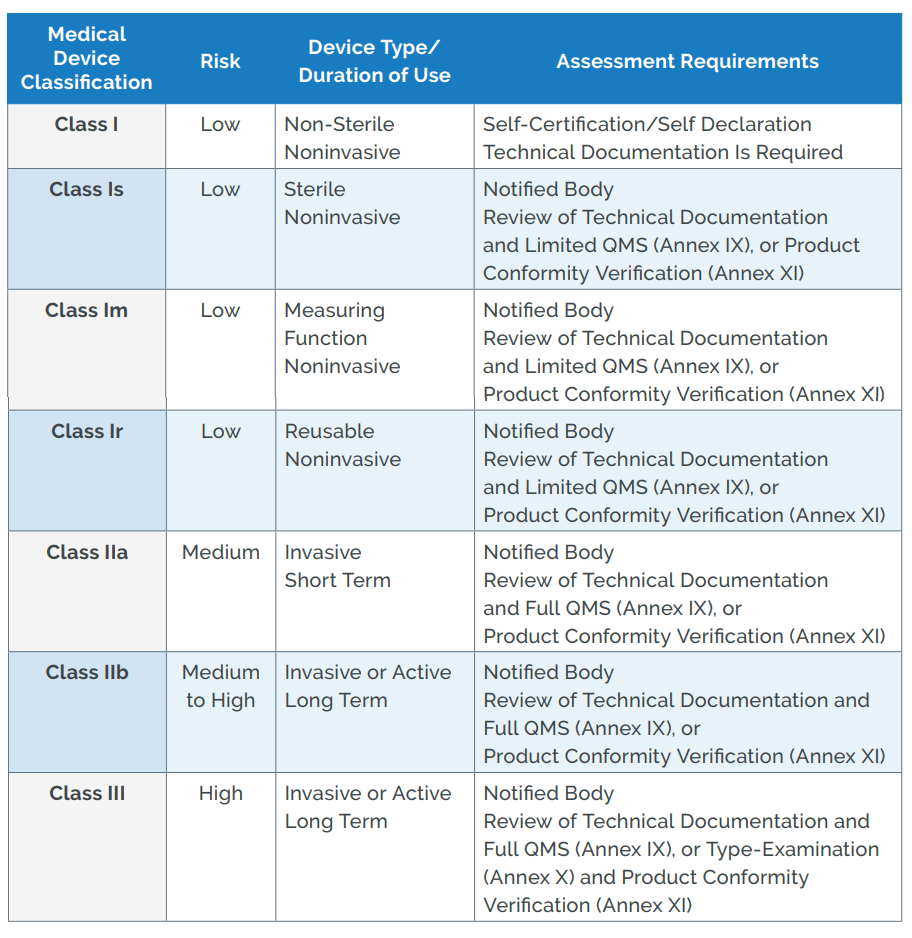
US FDA and EU Risk Classification of Medical Devices | Emergo by ULFDA and EU Medical Device Risk Classification: Impact on Human Factors